
- July 11, 2022
- by Lianeth Perez
- 0 Comments
Safety measures for the reprocessing of duodenoscopes
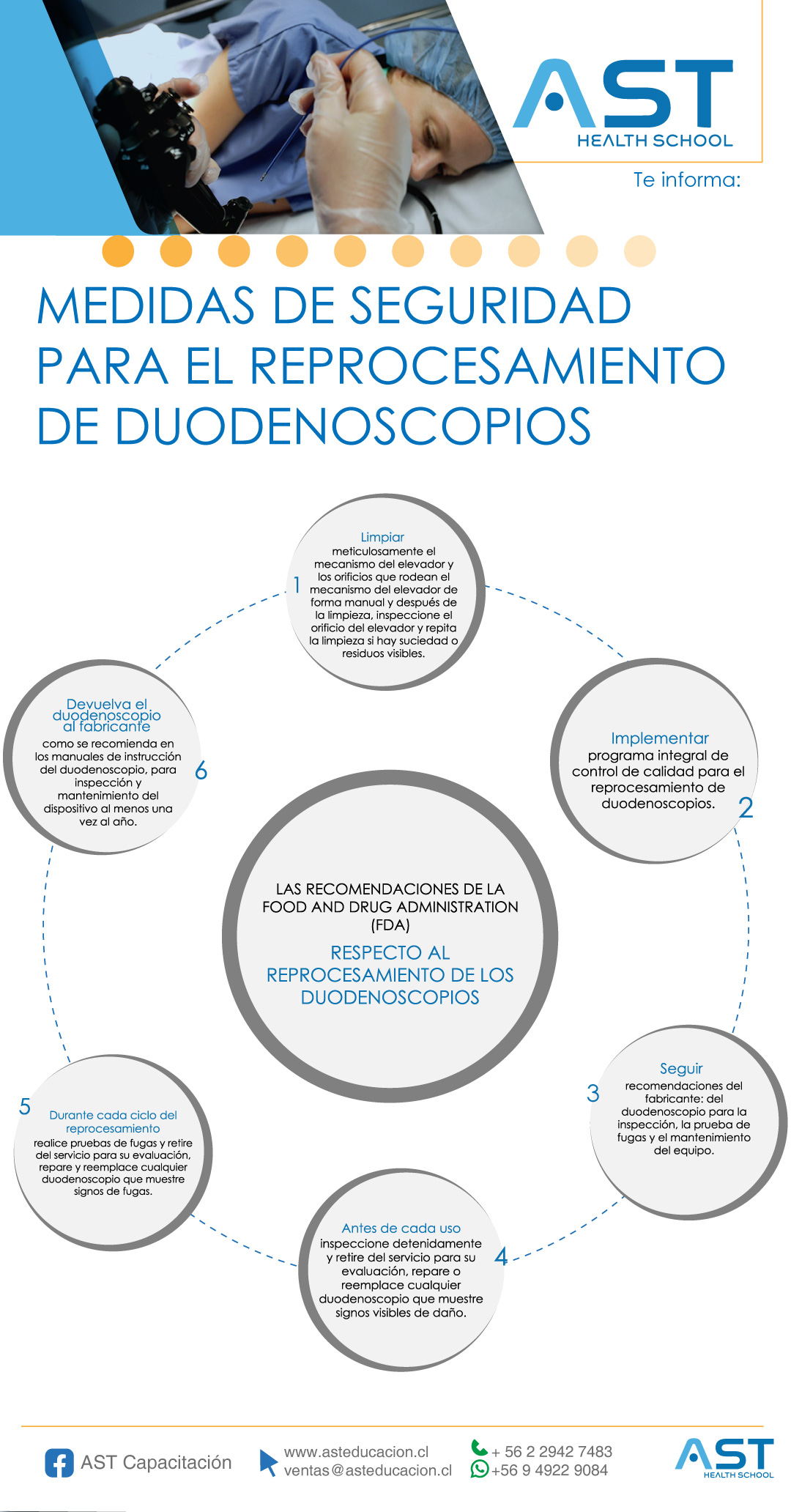
The Food and Drug Administration (FDA) in its safety communication provided a detailed list of supplementary measures for the reprocessing of duodenoscopes that can be implemented to reduce the risk of transmission of infections, such as: Microbiological culture, Ethylene Oxide Sterilisation, use of chemical sterilising liquid (HLD) processing system, repeat HLD process.
You can review the full infographic here: Safety measures for duodenoscope reprocessing
Relatest Posts
April 30, 2024
Wound Sutures
April 25, 2024
Diabetic foot disorders
April 24, 2024
Care Management in Vascular Ulcers
April 23, 2024
Hemodialysis HCAI prevention
April 18, 2024
Arterial Ulcers
April 17, 2024
Phases of healing
Categories
Recent Blogs
Wound Sutures
April 30, 2024Diabetic foot disorders
April 25, 2024Care Management in Vascular Ulcers
April 24, 2024